
A study led by Northwestern Medicine investigators has identified the molecular mechanisms within protein complexes that promote cell-to-cell adhesion and communication, according to findings published in the Proceedings of the National Academy of Sciences.
Sergey Troyanovsky, PhD, professor of Dermatology and of Cell and Developmental Biology, was senior author of the study.
Cadherins and catenins are proteins that form multiprotein complexes, helping bind cells together and stabilize cell-cell interactions, thereby forming different tissues. Those complexes, called cadherin-catenin complex (CCC), form clusters, but exactly how other CCC-associated proteins are recruited into these clusters and how they affect the overall clustering process has up until now remained understudied, according to Troyanovsky.
Using mass spectrometry and cross-linking, a proteomics approach which involves chemically “linking” two or more neighboring molecules by a covalent bond, the investigators discovered that most CCC-associated proteins interact with CCCs outside of adherens junctions, or protein complexes that include cadherin receptors. Furthermore, structural modeling revealed that there is limited space for CCC-associated proteins to form clusters in the first place.
Next, the investigators analyzed two example CCC-associated proteins essential for cell polarity and cell proliferation, and found that each protein formed separate CCC-associated clusters.
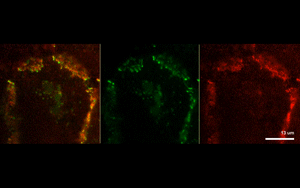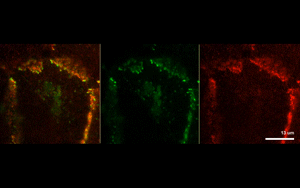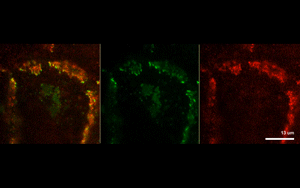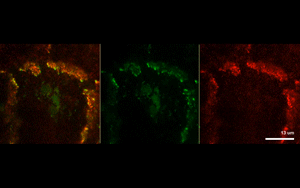
The findings suggest that protein-driven CCC clustering plays a role in cell-to-cell adhesion, as well as enables cells to communicate with each other by synchronizing their signaling networks.
“Different proteins which associate with CCC can sort cadherin into different populations of CCC clusters, and that’s important because it’s actually a mechanism of how signaling units can be equalized in different cells,” said Troyanovsky, who is also a member of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University.
Troyanovsky said the findings may also improve the understanding of the intracellular mechanisms that give rise to skin diseases, as most skin diseases are caused by defects in cell-to-cell adhesion within the epidermis.
The work was supported by National Institutes of Health grants AR44016 and AR057992.






