Northwestern Medicine scientists uncovered new insights into the molecular processes involved in the development of heart cells – findings that may lead to better strategies to repair heart tissue damaged from heart attacks and better understanding of congenital heart malformations.
The study, published in the journal Stem Cells, was led by MPH/PhD student Erin Lambers.
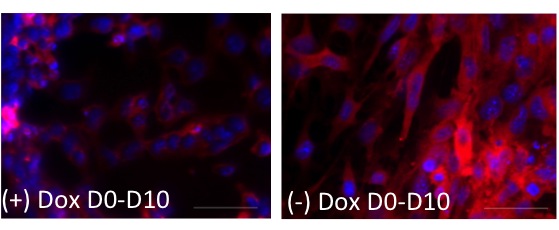
Scientists have known the transcription factor Foxc1 plays an important role in the development of cardiomyocytes, the heart cells that allow the heart to contract and relax, but the mechanisms that guide embryonic stem cells to become cardiomyocytes have been poorly understood.
The study identified the relationship between Foxc1 expression levels and the development of cardiomyocytes and how it affected cardiomyocyte function.
“There is a lot of research going on – on how to make these cardiomyocytes from embryonic stem cells. Modulating the genetics, you can increase the number, but this doesn’t necessarily mean they will be able to function properly,” said Lambers, who works in the lab of Tsutomu Kume, PhD, associate professor of Medicine in the Division of Cardiology and of Pharmacology.
In the current study, Lambers said, “we were able to show that not only could we create these heart cells and a lot more of them, but they could function normally by beating in synchrony with the heart cells around them.”

After altering Foxc1 expression levels, Lambers noticed a correlation between the amounts of the production of cardiomyocytes from stem cells. With lowered levels of Foxc1, there was a decrease in cardiomyocyte differentiation, the process of a cell changing from one cell type to another. If they raised levels of Foxc1, the scientists saw a similar increase in differentiation.
Lambers also tested the function of these cardiomyocytes, demonstrating their ability to respond to external electrical stimuli and beat in synchrony with neighboring cells by probing them with electric current and calcium handling techniques. The study also found cardiomyocytes with high levels of Foxc1 beat faster compared to cardiomyocytes with normal levels.
“You can visualize when a cardiomyocyte contracts and relaxes because during this process it uptakes calcium and releases it,” said Lambers. “So, by labeling calcium you can see these waves in response to electrical stimuli through their calcium handling.”
In addition, Lambers found Foxc1 enhances cardiomyocyte function in part by controlling another gene, myosin heavy chain 7, which is responsible for cardiomyocyte contraction.
Now that they have found one gene target of Foxc1, Lambers said she plans to use ChIP-sequencing, a method used to analyze protein interactions with DNA, to find other targets of Foxc1.
“In future studies, we will also want to see whether or not the cells we create can integrate into the heart and repair it after a heart attack,” she said.
The research was supported by National Institutes of Health grants R01 HL093439, R01 HL113541, R01 EY019484, R01 HL126920 and R25 GM079300 and American Heart Association pre-doctoral fellowship grant 15PRE25080006.






