
Northwestern Medicine scientists have developed a molecular “scaffold” capable of enhancing electrical activity and growth in neurons, which may prove useful in treating spinal cord injuries, according to recent results published in ACS Nano.
According to the National Spinal Cord Injury Statistical Center, 17,730 new spinal cord injuries are diagnosed each year, and an estimated 291,000 people live with spinal cord injuries in the U.S.
Injury to the central nervous system, including spinal cord injuries, often results in long-term nervous system dysfunction, as those neurons have limited ability to regenerate. The current study investigated new approaches to enhance that regrowth process, according to Samuel Stupp, PhD, the Board of Trustees Professor of Materials Science and Engineering, Chemistry, Medicine, and Biomedical Engineering, who was senior author of the study.
“Effective therapies for regeneration in the central nervous system are basically not available at the moment,” said Stupp, who is also the founding director of the Simpson Querrey Institute for BioNanotechnology (SQI) and its affiliated research center, the Center for Regenerative Nanomedicine. “There are some ideas around using stem cells and small-molecule drugs, but our approach is something very different. We have developed nanoscale fibers, formed by tens of thousands of molecules, with the capacity to signal neurons and other cells and built from naturally occurring building blocks that are completely safe to use. By design, the water-soluble nanofibers gel instantly into a scaffold-like structure upon injection at a tissue site where regeneration is needed. After a few weeks, the scaffold jump starts regenerative processes and then disintegrates into nutrients for the cells.”
Previous research has shown that certain proteins can be introduced to a spinal cord injury site to promote healing, but the proteins’ short half-lives prevent them from providing long-lasting results.
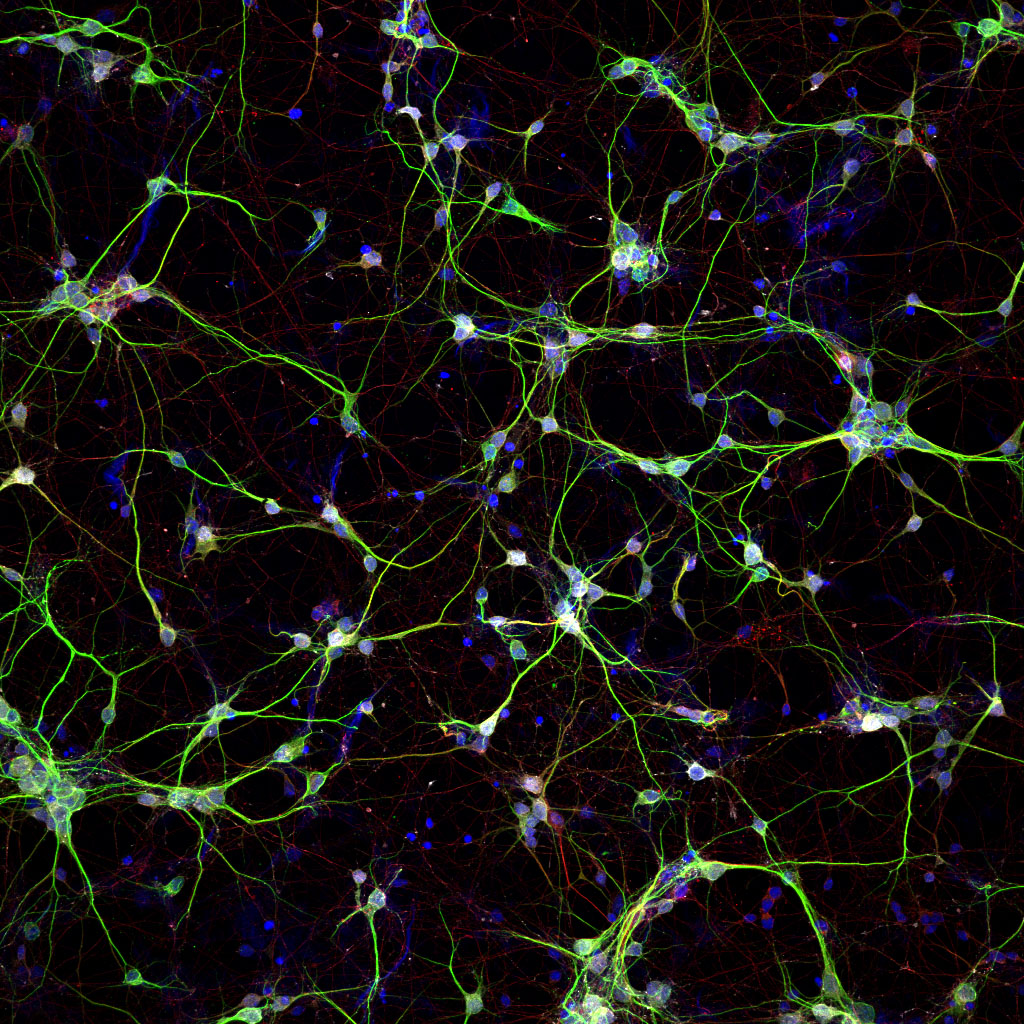
In the study, investigators sought to develop a novel type of nanofiber that mimics the bioactivity of the protein netrin-1 and could sustainably deliver signals to neurons over long periods of time. Netrin-1 is known to promote new neural connections and growth and could be instrumental in guiding axons — the long extensions of neurons that transmit electrical signals — to their targets to enable walking ability after spinal cord injury.
First, study investigators designed a peptide amphiphile, the type of molecule used in the Stupp laboratory to create bioactive nanofibers, that had a netrin-1 mimetic circular peptide attached to interact with a specific cell receptor. The netrin-1 mimetic peptide is extremely small compared to the protein and contains a key amino acid sequence that activated the targeted cell receptors for the desired bioactivity, according to the study.
When investigators exposed the nanofibers to cortical mouse neurons in vitro, they observed enhanced electrical activity and more outgrowth of neurites – key indicators of nerve regeneration. Protein analysis confirmed that the nanofibers activated neuronal netrin-1 receptors and successfully mimicked the protein over longer periods of time, according to the study.

“We saw that the netrin-1 mimetic peptide amphiphile nanofiber was able to be just as bioactive as the netrin-1 protein,” said Cara Smith, a biomedical engineering PhD candidate in the Stupp laboratory and first author of the paper. “Not only was it able to enhance neurite outgrowth, but it was also able to affect neuronal maturation and guide the development of new synapses, or points of communication between neurons.”
The Stupp lab has already completed a preliminary study evaluating the nanostructure’s healing abilities in live animals, with some promising early results, Stupp said.
“This nanofiber offers a vision for very potent therapies for regeneration in the central nervous system that are completely safe to use, are bioactive and very effective,” Stupp said. “They also can biodegrade safely once they have done their job. That sort of platform doesn’t exist right now.”
The study was supported by the Center for Regenerative Nanomedicine at the Simpson Querrey Institute for BioNanotechnology; National Institute of Biomedical Imaging and Bioengineering grant 5R01EB003806; grant R01NS104219 from the National Institute on Neurological Disorders and Stroke as well as the National Institute on Aging; and the New York Stem Cell Foundation.






