Orai1 calcium channels commonly found in immune cells are also present in the brain and regulate synaptic plasticity, according to a Northwestern Medicine study published in Cell Reports.
The presence of these channels in the brain opens up a new avenue of research for neurobiologists and establishes a new player in cognitive processing in the central nervous system, according to Murali Prakriya, PhD, professor of Pharmacology and senior author of the study.

“We were not sure what role these channels played in brain neurons,” Prakriya said. “The early view that these channels are present only in non-excitable cells is not true. We now know that they are highly expressed in the nervous system and are regulating the ability to learn and other higher-level cognitive tasks.”
Orai1 channels, named after the Horae who guarded the gates to Mount Olympus in Greek mythology, are calcium-selective ion channels. Previous studies from the Prakriya laboratory showed that these channels allow calcium to flow into T-cells, helping activate the body’s immune response.
Cells throughout the body employ calcium to control many cellular processes, including in dendritic spines, the small bulbous projections from neurons that receive synaptic activity from other neurons. However, the finer details of the pathways through which calcium enters dendritic spines to regulate synaptic plasticity are not well-understood, Prakriya said.
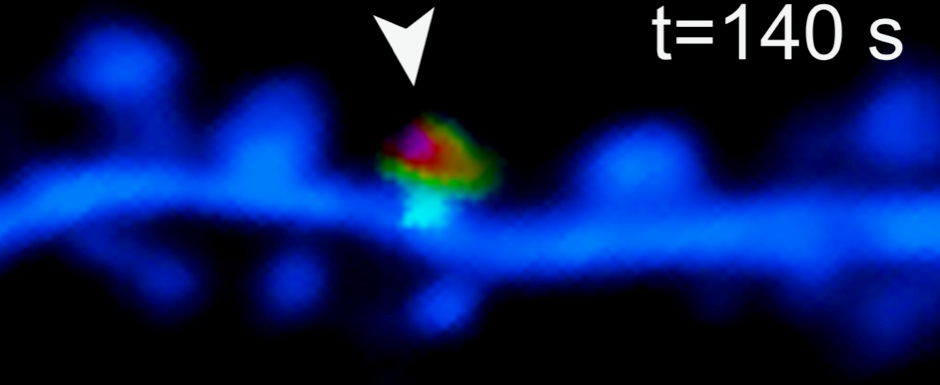
In the current study, investigators created modified neurons that lacked Orai1 channels. Using confocal microscopy and genetically encoded calcium dyes, they observed an almost complete loss of calcium influx activity in the dendritic spines of Orai1-deficient mice in response to synaptic stimulation. This was correlated with a loss of synaptic plasticity — the process by which neurons grow, change or develop stronger connections through activity — which is a crucial part of memory and learning.

“The changes in calcium signaling were extremely significant and larger than we expected ,” said Mohammad Mehdi Maneshi, PhD, a former postdoctoral fellow in the Prakriya laboratory and lead author of the study. “We conducted a wide range of additional control experiments to make sure we were looking at a true Orai1 deficiency phenotype.”
The investigators also created genetically modified mice that lacked Orai1 channels in excitatory neurons, but retained them elsewhere in the body and the brain. These mice exhibited mostly normal behavior, but their learning and memory was strongly impaired based on tests conducted by Maneshi and Anna Toth, a student in the Medical Scientist Training Program (MSTP) and a co-author of the study.
“Their locomotion was normal, they could eat normally, swim normally — but it was specifically in tests of long- and short-term memory where we saw deficits,” Toth said.
Working with Geoffrey Swanson, PhD, professor of Pharmacology and a co-author of the study, the scientists performed electrophysiological analysis of synaptic communication in Orai1-deficient mice. They found that baseline synaptic transmission was unaffected, but a process called long-term potentiation was almost completely blocked.
“Long-term potentiation is probably the best cellular correlate of learning and memory, and so the deficit in this process was very exciting and directly relatable to the cognitive deficits,” Prakriya said.

These findings represent a new area of inquiry for neurobiology, Prakriya said. While the channels are mostly similar to those seen in immune cells, there are important differences. In neurons, the study found that these channels are operating at much faster speeds when compared to immune cells, which makes sense given the pace of activity in the brain versus the immune system.
“There is likely some kind of molecular specialization that we have not yet identified that promotes rapid channel activation,” Prakriya said.
Further, Orai1 deficiencies bear a striking similarity to a class of as-yet unexplained intellectual disabilities caused by small deletions on chromosome 12 that encompass the region that codes for Orai1.
“There’s a strong possibility that these cases of unexplained impaired cognition may be traceable to Orai1 channel dysfunction,” Prakriya said.
Other co-authors of the study include Jelena Radulovic, MD, PhD, adjunct professor of Psychiatry and Behavioral Sciences; Richard Miller, PhD, the Alfred Newton Richards Professor of Pharmacology and of Psychiatry and Behavioral Sciences; and Megumi Yamashita, PhD, DDS, research assistant professor of Pharmacology.
Prakriya and Miller are members of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University.
This work was supported by National Institutes of Health grants R01NS057499, R01NS115508, R01NS080598 and F30 NS090817.