A new full-body scan could help clinicians to better assess toxicity during cancer treatment, according to a Northwestern Medicine study published in Clinical Cancer Research.
The scan, which detects the presence of molecules exposed during tissue damage, could give a precise evaluation of patient toxicity during chemotherapy, said Ming Zhao, PhD, associate professor of Medicine in the Division of Cardiology and senior author of the study.
“After a single chemotherapy treatment, you already see changes,” said Zhao, who is also a member of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University. “This could give doctors the opportunity to intervene and reduce the dose or switch to another drug for example, hopefully preventing any further damage to the patient.”
Steven Johnson, ’18 PhD, postdoctoral fellow in the Zhao laboratory, was lead author of the study.
Chemotherapy works by targeting and killing actively dividing cells. While this fights cancer, casting such a wide therapeutic net damages healthy cells as well, resulting in potentially harmful side effects in patients.
While there are a range of clinical tools to measure cancer’s response to chemotherapy, tools that assess patient toxicity are limited, according to Zhao.
“While tumor kill is the main therapeutic goal in anticancer treatment, toxicity is equally important because it tends to dictate the patients’ tolerance ceiling for treatment,” Zhao said. “Current tools rely on patient symptoms and subsequent blood or serum tests, which can lag behind actual tissue damage. Instead, an imaging based method could be a quicker and more accurate way to ascertain patient toxicity.”
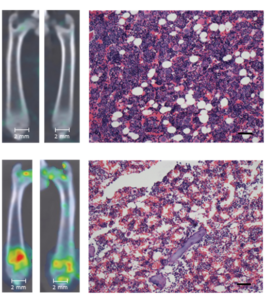
In the current study, scientists mapped tissue damage in rodent models using a novel whole-body imaging technique developed in collaboration with the Chemistry of Life Processes Institute’s Center for Advanced Molecular Imaging (CAMI). The technique detected a lipid molecule that is not accessible in normal cells, but becomes visible in dead and dying cells.
The lipid molecule, called phosphatidylethanolamine (PE), usually resides inside the cell. However, when a cell dies, it deactivates enzymes that maintain the asymmetrical structure of the cell membrane, redistributing PE to the cell surface.
“This provides a molecular marker for detecting cell death,” Zhao said.
The scientists compared the results of blood and serum tests to the imaging results, finding the signal changes in the scans correlated with the results of the conventional tests — with the scan providing earlier, broader and more dynamic information, according to Zhao.
“Damage to the skin can be highly local; if you take a biopsy, you might miss it,” he said. “With this test, you look at the entire organ; if there is a heterogeneous distribution of damaged tissue, you can tell where it went wrong.”
They also tested the scan in both male and female rodents, finding it detected damage in reproductive organs, a life-changing issue for some patients.
“Reproduction is a major issue in younger patients,” Zhao said. “This may help predict if this individual will have problems down the line.”
Further, the scan could also detect damage, or lack thereof, in the tumor itself, providing another data point to drive clinical decision making.
“A lack of PE on the tumor will tell you that the drug isn’t killing the tumor effectively, suggesting you should make the decision to switch to another treatment,” Zhao said. “This scan should help optimize cancer treatment to maximize tumor damage and minimize side effects.”
Now, Zhao and his collaborators are exploring the technology’s potential in humans.
“We’ve shown the proof of concept; this can be done,” Zhao said. “But for human translation, we need to make sure everything works well — and safely.”
Currently, the scientists are working on implementing a second-generation imaging agent, which could produce significantly greater data quality for clinical translation.
The study was an inter-departmental and interdisciplinary effort, involving authors from different areas of expertise. Co-authors included Chad Haney, PhD, research associate professor of Chemistry of Life Processes Institute, Biomedical Engineering, and of Radiology; Gennadiy Bondarenko, PhD, postdoctoral fellow; Emily Waters, research associate at the CAMI; Andy Tran, research assistant; Thomas O’Halloran, PhD, professor of Medicine in the Division of Endocrinology, Metabolism and Molecular Medicine. Former Feinberg faculty Raymond Bergan, MD; Andrew Mazar, PhD; Andrey Ugolkov, MD, PhD and Lin Li, MD, were also co-authors.
This work was funded by a “Provocative Questions” grant from the National Cancer Institute (NCI) R01 CA185214 awarded through the Chemistry of Life Processes Institute, 1S10OD016398 to fund acquisition of the SPECT/CT scanner in CAMI, and 5R01HL102085 and NCI CCSG P30 CA060553.