
Northwestern Medicine investigators contributed to the development of a model of synapse-organizing protein structures to reveal how another protein, called MDGA, can interfere with the function of synapses, which may play a role in neurological disorders including autism spectrum disorders and schizophrenia.
Jeffrey Savas, PhD, assistant professor of Neurology in the Division of Movement Disorders, and Savas lab member Samuel Smukowski were co-authors on the paper published in Neuron.
When a synapse is created, the proteins neuroligin (NL) and neurexin (NRX) “shake hands,” creating cell adhesion and ensuring proper signal transmission. Dysfunction in NL-NRX signaling is associated with psychiatric and neurodevelopmental disorders, and MDGA proteins are known to play a role in interrupting healthy synaptic connections, according to the study.
“In this study we used a biochemical method that we previously developed to identify a collection of new trans-synaptic adhesion interactions,” Savas said. “We are currently researching the role of specific synaptic adhesion protein interactions at distinct brain synapses in a variety of neurological conditions.”
The investigators used diffraction-quality crystals and X-ray scattering to create detailed structural models of MDGA1, NL1-MDGA1 complex and a spliced NL1 isoform. In models with poor NL-NRX signaling, they found the existence of two large MDGA molecules cradling NL, shielding it from interacting with NRX in an unexpected arrangement.
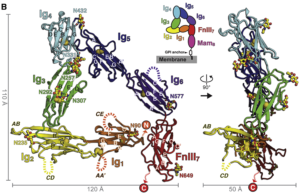
The authors also found a new NL-NRX interaction site, named Site II, highlighting the versatility of NL to fold and accommodate new interactions.
The study’s findings highlight the potential of modulating NL-NRX interactions throughout the brain. These interactions could affect the overall balance of excitatory and inhibitory synaptic transmission, a disease feature associated with some autism spectrum disorders.
While it’s important to understand the molecular mechanism behind poor NL-NRX signaling, the key to treating or preventing the aberrant structures is in the genetics that produce them, according to the authors.
“Our findings considerably broadened this interaction network beyond that previously envisioned,” the authors wrote. “Moreover, our structural studies constitute an essential guide toward the generation of directed therapies targeting these gene products to restore excitatory-inhibitory balance.”
Savas is also an assistant professor of Medicine and of Pharmacology.
This work was funded by the UK Medical Research Council grants G0700232, L009609, and MC_UP_1201/15; Canadian Institutes of Health Research grants FDN-143206; ERA-NET NEURON 2015 Cofund Program under Horizon 2020; National Institutes of Health (NIH) grant R00 DC013805; European Molecular Biology Organization grant ALTF 1116-2012; Marie-Curie FP7-328531 long-term postdoctoral fellowships; CIHR 338646; Michael Smith Foundation for Health Research 16005 postdoctoral fellowships; Cancer Research UK grant C375/A10976; Nuffield Department of Medicine Prize Student; NIH grants MH085926, GM105730 and F32NS093753; Canada Research Chair; Medical Research Council senior research fellow; Wellcome Trust Centre for Human Genetics is supported by Wellcome Trust grant 090532/Z/09/Z.






