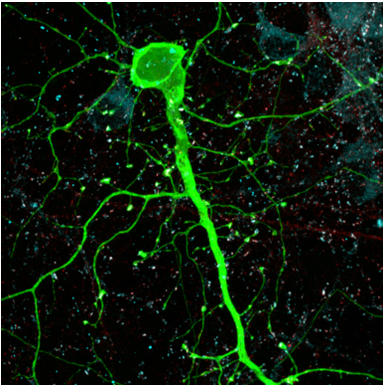
Northwestern Medicine scientists have demonstrated a new method of analyzing non-coding regions of DNA in neurons, which may help to pinpoint which genetic variants are most important to the development of schizophrenia and related disorders.
Peter Penzes, PhD, the Ruth and Evelyn Dunbar Professor of Psychiatry and Behavioral Sciences, was a lead author of the study, published in the journal Cell Stem Cell. Marc Forrest, PhD, a post-doctoral fellow in Penzes’ laboratory, was the first author.
Over the last decade, large genetic studies have identified thousands of genetic variants associated with mental disorders. Most of these risk variants, however, are found within non-coding regions — parts of DNA that do not encode for proteins — whose function in disease development has been poorly understood.
“Ten years ago, there was very little known about the genetic basis of mental disorders like schizophrenia. Now the problem is the opposite: we have too many genes,” Penzes said. “Studying each variation one by one — and how it contributes to actual disease — is difficult, so methods to reduce that number can be very useful. And that’s what this study did.”
The scientists demonstrated that by mapping out open chromatin regions in neurons derived from human stem cells, they could identify active non-coding DNA that contain a key subset of psychiatric risk variants that are most relevant to disease.
While the model was demonstrated in schizophrenia, the same method could be applied to other mental disorders as well, such as autism spectrum disorders or bipolar disorder.
Developing such a technique is critical to help scientists in the field concentrate their efforts on investigating the most important variants.
The findings also deepen the overall understanding of how such non-coding regions affect disease.

As a case study, the scientists used the new model to analyze thousands of risk variants that have been associated with schizophrenia and narrowed it down to a small list of key variants, of which they chose one to investigate.
They then used the gene-editing tool CRISPR to alter that risk variant into a variant not associated with disease, and demonstrated that the change had an effect on the connectivity of the cell, suggesting it played a role in neurodevelopment.
“In the past, these non-coding regions have been called ‘junk DNA’ because there was this misconception that they had no function,” Forrest said. “With this kind of technique, we’re starting to understand how non-coding regions can affect disease risk, even if they have more indirect roles than the actual protein-coding regions.”
In the future, the model using human neurons from induced stem cells could also serve as a valuable tool to screen potential drugs for such disorders, and discover which ones result in changes in the neuronal phenotype, Penzes said.
Penzes is also a professor of Physiology and of Psychiatry and Behavioral Sciences.
The study was supported by National Institutes of Health (NIH) grants R21MH102685, R01MH106575 and R01MH097216; a Swiss National Science Foundation early postdoctoral mobility fellowship; and grant F31NS084551.