In early March, when much of the world came to a halt because of the COVID-19 pandemic, medical practice and medical education were disrupted like never before. Within days of Illinois’ stay-at-home order taking effect, Feinberg faculty rapidly transitioned to an almost completely virtual medical curriculum, requiring students to quickly adapt and pursue this “new normal” without hesitation.
The pandemic’s impact on Feinberg’s research enterprise was no different: Projects deemed essential required some investigators to socially distance in laboratories and facilities, while others worked from home to analyze data, devise new experiments and conduct meetings with colleagues virtually. Many investigators transitioned their ongoing research to COVID-19, others had to rethink the execution of their clinical trials in order to abide by social distancing guidelines, and some working in wet laboratories were forced to temporarily stop their research altogether.
Despite these incredible challenges and obstacles, Feinberg kept moving onward and upward. In the 2019-2020 fiscal year, Feinberg principal investigators secured a record-breaking $643 million in research funding and awards — an impressive 20 percent increase over the previous year. Of this total, more than $24 million in awards were awarded to Feinberg investigators for COVID-19 related research.
January: AI Model Improves Breast Cancer Detection
An international team of investigators, including those from Northwestern Medicine, developed a new artificial intelligence (AI) model that predicted breast cancer in mammograms more accurately than radiologists, reducing false positives and false negatives. The study, published in Nature, was co-authored by Mozziyar Etemadi, MD, PhD, research assistant professor of Anesthesiology and of Biomedical Engineering at the McCormick School of Engineering. “Breast cancer is one of the highest causes of cancer mortality in women,” Etemadi said. “Finding cancer earlier means it can be smaller and easier to treat. We hope this will ultimately save a lot of lives.”
February: Uncovering the Cellular Mechanisms Behind Genetic Mutations in ALS
A team of Northwestern Medicine scientists led by Evangelos Kiskinis, PhD, assistant professor in the Ken and Ruth Davee Department of Neurology, discovered that mutations in the largest genetic contributor to ALS leads to the dysfunction and eventual degeneration of motor neurons in the brain.
Published in Neuron, the findings shed light on the mechanisms and consequences of a defect in a gene called C9orf72, which disrupts the localization of proteins involved in RNA and protein metabolism. The study may also aid the development of novel therapeutic interventions for patients with the neurodegenerative disease, which currently has no cure.
March: New Drug Target Found for COVID-19
Northwestern Medicine investigators were charged with helping to widen the scope of research about the novel SARS-CoV-2 virus. Led by Karla Satchell, PhD, professor of Microbiology-Immunology and director of the Center for Structural Genomics of Infectious Diseases, a team of scientists identified a potential drug target in SARS-CoV-2: two critical proteins in a complex called nsp10/16. According to the investigators, a drug that can inhibit nsp10/16 would allow the immune system to detect the virus and eradicate it faster.
“This is a really beautiful target, because it’s a protein absolutely essential for the virus to replicate,” Satchell said. The investigators also mapped the atomic structure of nsp10/16, which was published for public use on the RSCB Protein Data Bank.
April: First Trial Shows Benefit for Genomically Targeted Prostate Cancer Treatment
In a first-of-its-kind clinical trial, advanced prostate cancer was treated based on its genomic makeup. The treatment delayed progression for patients with metastatic castration-resistant prostate cancer, a deadly and treatment-resistant form of the disease.
Patients randomly selected to receive the drug olaparib experienced delayed disease progression for seven months on average compared to just three months for the standard treatment cohort. Additionally, about 60 percent of men in the olaparib group showed no disease progression at six months compared to 23 percent in the standard cohort. The clinical trial, published in the New England Journal of Medicine and led by Maha Hussain, MBChB, the Genevieve E. Teuton Professor of Medicine in the Division of Hematology and Oncology, represents a breakthrough in treating this deadly cancer and for precision medicine more broadly.
May: Monitoring COVID-19 from Hospital to Home: First Wearable Device Continuously Tracks Key Symptoms
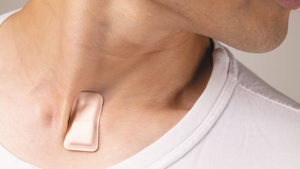
Investigators at Northwestern University and the Shirley Ryan AbilityLab developed a novel wearable device to detect early signs and symptoms associated with COVID-19. The device, no larger than a postage stamp, can be worn 24/7 and continuously measures and interprets coughing and respiratory activity through custom-made AI algorithms. John Rogers, PhD, professor of Neurological Surgery and the Louis Simpson and Kimberly Querrey Professor of Materials Science and Engineering, Biomedical Engineering and Neurological Surgery at the McCormick School of Engineering, led the technology development. Arun Jayaraman, PhD, associate professor of Physical Medicine and Rehabilitation, of Medical Social Sciences and of Physical Therapy and Human Movement Sciences, led the AI algorithm development efforts. “We hope, and we believe, that these devices may help in these efforts by identifying and quantifying characteristics and essential features of cough and respiratory activity associated with this disease,” Rogers said.
June: Cancer Cells Hijack Nucleotide Metabolism to Boost Cell Proliferation
Issam Ben-Sahra, PhD, assistant professor of Biochemistry and Molecular Genetics, was the lead author of a study published in Molecular Cell, which explored how genetic mutations associated with many cancers drive proliferation. Specifically, the team of investigators found that mutations in the genes RAS and RAF allow cancer cells to create their own nucleotides, one of the molecular building blocks of cells throughout the body. Although this molecular connection helps cancers spread, it reveals an opportunity for therapeutic intervention. “Now that we’ve identified this molecular connection, we can imagine targeting this pathway. This could be one of the Achilles’ heel of the cancer,” said Ben-Sahra, who is also a member of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University.
July: COVID-19 Complicates Cancer Treatment
Cancer patients with other comorbidities have a higher risk of dying from complications due to COVID-19 and should discuss the risks and benefits of continuing cancer treatment with their physician, according to a recent published in The Lancet. The study was co-authored by Alicia Morgans, MD, MPH, associate professor of Medicine in the Division of Hematology and Oncology; Mary Mulcahy, MD, ’00 GME, professor of Medicine in the Division of Hematology and Oncology; and Firas Wehbe, MD, PhD, associate professor of Preventive Medicine in the Division of Health and Biomedical Informatics and chief research informatics officer. The findings were derived from clinical data collected over 30 days from more than 900 patients receiving cancer treatment at 100 different participating institutions.
August: AI-Enhanced Approach Offers New Hope for Earlier Autism Diagnoses
A team of investigators from Northwestern Medicine, Ben Gurion University, Harvard University and the Massachusetts Institute of Technology developed a novel precision medicine technique that uses AI to identify a subtype of autism by overlaying an array of biomedical and healthcare data. The technique, detailed in a study published in Nature Medicine, successfully identified through elaborate data analysis dyslipidemia-associated autism, which represents 6.55 percent of all diagnosed autism spectrum disorders in the United States. “Today, autism is diagnosed based only on symptoms, and the reality is that when a physician identifies it, it’s often that early and critical brain developmental windows have passed without appropriate intervention. This discovery could shift that paradigm,” said Yuan Luo, PhD, associate professor of Preventive Medicine in the Division of Health and Biomedical Informatics and co-first author of the study.
September: Biological Sex Affects Genes for Body Fat, Cancer, Birth Weight
Barbara Stranger, PhD, associate professor of Pharmacology, led a team of investigators that found biological sex influences gene expression in almost every type of human tissue. The study, published in Science, reports that these sex differences are observed for genes involved in many functions, including how people respond to medication, how women control blood sugar levels in pregnancy, how the immune system functions and how cancer develops. Furthermore, Stranger said that these findings underscore the importance of considering sex as a biological variable in human genetics and genomics studies.
October: Utilizing B-Cells to Promote Glioblastoma Immunity
Northwestern investigators developed a novel vaccine that utilizes a specialized group of B-cells to promote anti-tumor immunity against glioblastoma, findings published in the Journal of Experimental Medicine. The vaccine, which is still in pre-clinical stages, is the first of its kind and may be an alternative to currently available immunotherapeutic approaches to treat the fatal brain cancer. Catalina Lee Chang, PhD, research assistant professor of Neurological Surgery, was the first author of the study and Maciej Lesniak, MD, chair and the Michael J. Marchese Professor of Neurosurgery, was the senior author. “With this vaccine, we are targeting the dual functionality of B-cells to tackle tumor immunosurveillance escape,” Lee Chang said. “We aim to utilize both the cellular and humoral immunity of B-cells.”
November: New Path Forward for Pancreatic Cancer Treatment
A study led by Mazhar Adli, PhD, associate professor of Obstetrics and Gynecology in the Division of Reproductive Science in Medicine, found that combining the current standard-of-care chemotherapy drug with a genetic inhibitor may improve treatment for pancreatic ductal adenocarcinoma (PDAC).The study was published in Proceedings of the National Academy of the Sciences (PNAS). There are currently no targeted treatments for PDAC and chemotherapy comes with a host of side effects, so this combination drug strategy is promising. “Pancreatic cancer patients are desperately awaiting a new drug combination,” Adli said. “We are hoping that we can initiate a clinical trial in collaboration with these drug companies as soon as possible.”
December: Scientists Unravel Zebrafish Epigenome
Northwestern Medicine investigators identified tissue-specific epigenetic regulators in zebrafish, filling in a longtime gap in the understanding of the organism’s genome. According to Feng Yue, PhD, the Duane and Susan Burnham Professor of Molecular Medicine and senior author of the study published in Nature, the findings could help scientists use the model organism to unravel human diseases such as cancer, since more than 70 percent of protein-coding genes in the zebrafish genome are also present in humans. “If you see something in the human genome and can’t establish a cause, you can use this data to perform experiments in zebrafish. This will be very valuable for researchers in this field,” said Yue, who is also the director of the Center for Advanced Molecular Analysis at the Institute for Artificial Intelligence in Medicine.






