Studying SARS-CoV-2, the virus that causes COVID-19, is just as important as studying how the body can develop an immune response to it, according to Elizabeth McNally, MD, PhD, the Elizabeth J. Ward Professor of Genetic Medicine and director of the Center for Genetic Medicine, who discussed findings from the ongoing Northwestern Medicine Screening for Coronavirus Antibodies in Neighborhoods (SCAN) study during a recent webinar.
The SCAN study, of which McNally is a co-investigator, utilizes an at-home COVID-19 antibody test to help investigators determine how many people have been exposed to SARS-CoV-2 and have developed antibodies, with the ultimate goal of learning whether these antibodies protect people against re-infection.

Behind the SCAN study and the development of the COVID-19 antibody test is a multidisciplinary team of Northwestern Medicine investigators, including McNally; Thomas McDade, PhD, the Carlos Montezuma Professor of Medical Social Sciences and of Anthropology at the Weinberg College of Arts and Sciences; Alexis Demonbreun, PhD, assistant professor of Pharmacology; Brian Mustanski, PhD, professor of Medical Social Sciences, Psychiatry and Behavioral Sciences, and director of the Institute for Sexual and Gender Minority Health and Wellbeing (ISGMH); Richard D’Aquila, MD, director of the Northwestern University Clinical and Translational Sciences (NUCATS) Institute, the Howard Taylor Ricketts, MD, Professor of Medicine in the Division of Infectious Diseases and associate vice president of research; and Nanette Benbow, MA, research assistant professor of Psychiatry and Behavioral Sciences.
McNally outlined the three phases of SCAN: antibody test validation; SCAN Chicago, which involved COVID-19 antibody testing of participants from more than 10 different zip codes across the city Chicago; and SCAN FSM, which involved COVID-19 antibody testing of Feinberg students, faculty and staff.
In March, McNally became involved with COVID-19 antibody research when she and her team in the Center for Genetic Medicine realized the need for antibody testing in the community. They also recognized that the study might be limited by participants not wanting to come to a health care facility during the pandemic.
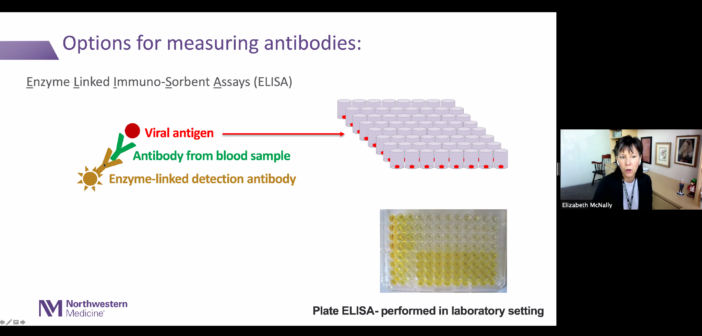
One month later, McNally and Demonbreun, in collaboration with McDade on Northwestern’s Evanston campus, developed testing protocols to detect SARS-CoV-2 antibodies using an enzyme-linked immunosorbent assay (ELISA). The platform was specifically designed to detect immunoglobulin G (IgG) antibodies against the receptor-binding domain of the SARS-CoV-2 spike protein, which is located on the surface of the viral cells and helps the virus infect normal cells. This, according to McNally, motivated the team to develop a test that specifically detects these antibodies.
“Having IgG antibodies to this receptor binding domain makes it more likely that those are going to be neutralizing antibodies, in that a neutralizing antibody may block the interaction of the spike protein with the receptor on surfaces themselves,” McNally said.

To expand antibody testing into the community, the test was built on McDade’s novel dried blood spot collection method, which requires drops of blood collected from a finger prick to be put on designated filter paper. The advantage of this test, according to McNally, was that it could be safely done at home and results could be sent through the mail, an ideal situation during a pandemic.
In May, investigators began enrolling select participants for the SCAN Chicago study from 10 different “pairs” of zip codes across the city: 60645 (West Rogers Park), 60660 (Edgewater), 60639 (Belmont Cragin), 60647 (Logan Square), 60612 (Near West Side), 60622 (Ukrainian Village), 60609 (Back of the Yards), 60615 (Kenwood), 60643 (Beverly), and 60655 (Mt. Greenwood).
Participants were asked to enroll for the study and be screened online, where they could then register and have an antibody testing kit mailed to their home address. So far, almost 5,500 kits have been sent out to study participants across the city, according to McNally.
“Part of why we pick these zip code pairs is because the rates of COVID positivity in these pairs were pretty different from each other, and we asked whether the serology was different in these neighborhoods paralleling to what was we were seeing based on the COVID testing,” McNally said, adding that the SCAN study is concentrating on recruiting diverse participants so that the study reflects Chicago’s current demographics.
Expanding their reach even further, the investigators launched SCAN FSM in July to determine antibody positivity in the Feinberg community. McNally said the study was also launched to determine if antibody levels were different in those who dropped off test kits in-person versus those who stayed at home and mailed in their results as seen in SCAN Chicago.
For SCAN FSM, antibody testing kits were distributed to participants and returned on site in the lobby of the Simpson Querrey Biomedical Research Center at scheduled times to decrease crowding. Of the 5,500 individuals invited to participate, more than 3,000 antibody testing kits have been completed by Feinberg students, postdoctoral students, fellows, staff and faculty, McNally said.
So far, McNally’s team has analyzed samples from the first 1,545 participants of SCAN that were collected during the summer, with the results being an equal mix between SCAN Chicago and SCAN FSM samples, McNally said.
According to McNally, results showed a similar exposure to COVID-19 in SCAN Chicago samples and SCAN FSM samples, with just under 20% of people having antibodies to the virus. The majority of these participants also had minimal to no symptoms and for those who had known COVID-19, 97% of these individuals will have a positive antibody test.
The team also had the opportunity to analyze samples from June through late August, finding that 75 percent of individuals who had SARS-CoV-2 antibodies continued to have antibodies 120 days after they took the test. The study hopes to sample participants again, about six months after the first test, to see if the antibody status changes. Also, they plan to see whether having antibodies makes people less likely to develop COVID-19.
McNally said her team is now seeing a few cases of participants who previously tested antibody positive develop COVID-19, which suggests that reinfection can happen. Knowing the health status of participants over time will potentially help guide strategies to mitigate COVID-19, according to McNally.
“This is going to be an ongoing study and it’s not going to go away anytime soon. As vaccines rollout, I think it’s going to be really important to see how health status changes, because a vaccine will just be one part of what we’re doing and there’s still going be endemic virus for some period of time,” McNally said.
Additionally, McNally said that the study is now entering its fourth phase: SCAN NUgene. For SCAN NUgene, investigators will analyze genomic data from the NUgene biobank, which houses over 14,000 DNA samples and electronic medical data from Northwestern Medicine patients, to identify genomic signals for COVID-19 susceptibility.
“We think this is a powerful platform for understanding some of the asymptomatic exposure in seropositivity, as well as some of the symptomatic positivity that’s happened out there,” McNally said. “It’s critical to have a diverse sample, not just for the social determinants of health, but also for this genomics component, to make sure that we’re really representing what our population looks like.”
This webinar was part of the Translational Applications in Public Health webinar series sponsored by the Institute for Public Health and Medicine (IPHAM) and the NUCATS Institute.






