Northwestern Medicine scientists have discovered a potentially new approach to treating a rare but devastating bone marrow disorder.
Fanconi anemia (FA) is an inherited blood condition caused by a mutation in a cluster of proteins responsible for DNA repair.
“These patients develop anemia – a decrease in red blood cells – low white blood cell counts and bone marrow failure, usually in late childhood,” said Elizabeth Eklund, MD, professor in Medicine-Hematology/Oncology.
The average life expectancy of an individual with the disorder, which occurs once in every 350,000 births, is 20-30 years. Children with Fanconi anemia who do not experience bone marrow failure frequently develop acute myeloid leukemia in adolescence or as young adults.
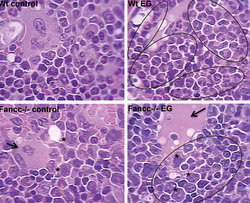
What Liping Hu, a postdoctoral fellow, and others in Dr. Eklund’s lab uncovered is that when mice with the disorder were asked to fight off infectious or inflammatory challenges, the result was an acceleration of bone marrow failure and leukemia.
The findings were published in the Journal of Clinical Investigation.
From left: Elizabeth Eklund, MD, professor in Medicine-Hematology/Oncology, and postdoctoral fellow Liping Hu recently published an article in the Journal of Clinical Investigation that may change the way Fanconi anemia is treated.
“This study suggests that protecting Fanconi anemia patients from repeated infections might delay bone marrow failure or progression to leukemia,” said Dr. Eklund, a member of the Robert H. Lurie Comprehensive Cancer Center of Northwestern University. “Currently, these patients are given bone marrow stimulating growth factors when they develop infection and our data suggests that these growth factors may be a part of the problem.”
Although FA is a blood disorder, it can also affect many of the body’s organs, tissues and systems. Children who inherit FA are at higher risk of birth defects and the disorder increases the chances of cancer and other serious health problems.
“When Liping Hu presented this work at the annual Fanconi Anemia Research Fund meeting in Houston this fall, there was a lot of interest because it suggests a major change in the treatment of patients,” Dr. Eklund said. “Hopefully, this publication will lead to clinical trials to determine if our studies translate well to humans.”
The work was supported by a research grant from the Fanconi Anemia Research Fund, VA Merit Review and National Institutes of Health grant HL088747.